What is Artificial Cervical Disc Replacement?
Degenerative spinal disease can affect the structure and integrity of the intervertebral discs that lie between the cervical vertebra. This can cause pain on neck movement, and can compress and irritate the nerves that originate from their and supply the arms. In such situations, the cervical disc may need replacing, and this is where anterior cervical disc replacement is conducted.
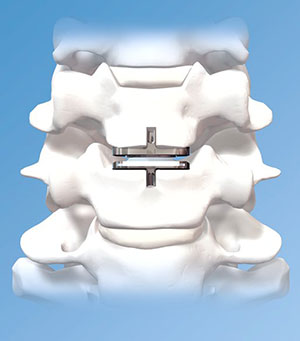
A popular device used to replace the degenerated disc is the ProDisc-C artificial disc.
Indications
ProDisc-C artificial cervical disc replacement is offered to patients with severe disc herniation or with degenerative disc disease. Normally, in such patients, spinal fusion is offered as a sole procedure, but given the loss of neck mobility that accompanies this, cervical disc replacement is now being offered a lot more.
The procedure
Once consent is obtained and anesthetic agent administered, the patient is placed in position and the area to be operated is cleaned with antiseptic solution. An incision is made on the front of the neck, and the tissues visualized are dissected in order to make room to access the cervical vertebra.
The diseased disc is removed and the area is shaped to allow the device to fit in snugly. Special instruments allow the surgeon to accurately create notches, or "keel" cuts, to hold the artificial disc in place.
The ProDisc-C artificial disc system is inserted in the space that is created. The disc is fitted to the vertebra above and below, and the device can now move similar to the neck's natural motion.
Once the device is in place, the incision is closed and the patient is discharged after a period of observation.
Benefits
The benefit of anterior cervical disc replacement using the ProDisc-C device is that the movement of the cervical spine is preserved completely. No spinal fusion is necessary. Patients experience a great deal of relief of symptoms, and a significant improvement in function. Patients can return to their normal daily activities a lot quicker than other surgeries. Device failures are rare.
Disadvantage
The ProDisc-C system cannot be used for patients with an allergy to titanium, polyethylene or other materials contained in disc implant. It should not be implanted in patients with an active infection.
Risks
These are rare but recognized. Bleeding, bruising and infection may be seen. Patient may sustain inadvertent nerve injury which can worsen pain. Blood clot formation can occur which requires treatment. Patients may develop difficulty swallowing and a change in their speech. All these complications and risks are extremely rare when the procedure is performed by expert surgeons.
